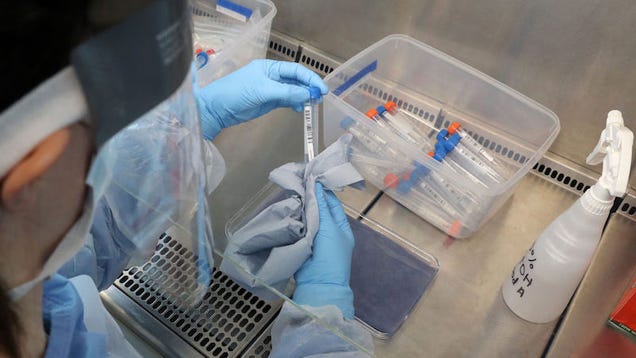
The Food and Drug Administration (FDA) on Saturday announced that it had issued an emergency use authorization for an at-home coronavirus sample collection kit. The kit allows consumers to self-collect a nasal sample and then send it off to be tested for the novel coronavirus, which causes the disease covid-19.
